Putting the elements in any kind of order would prove quite difficult. (Image credit: Oxford Science Archive/Print Collector/Getty Images)
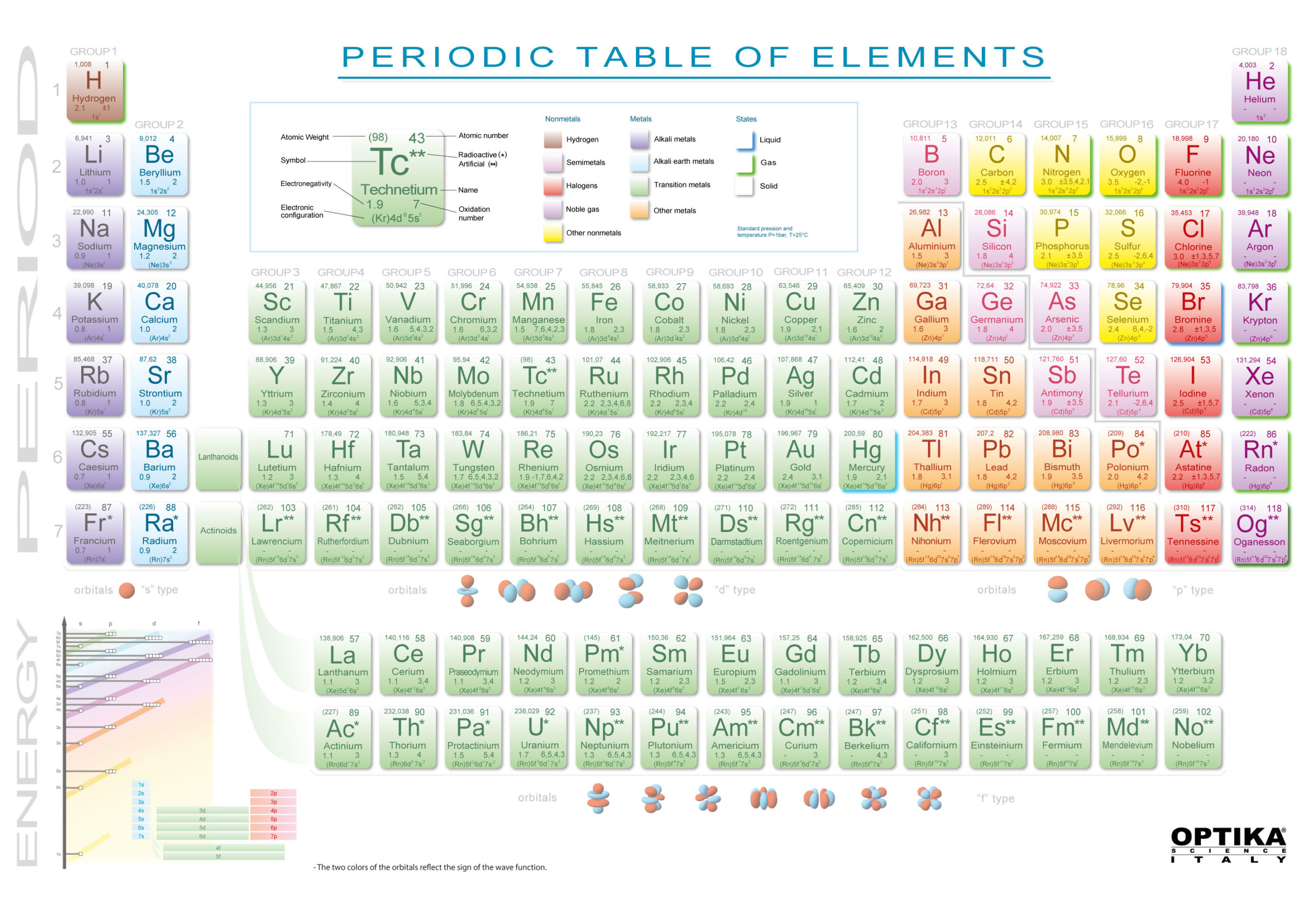
For instance, all the group 18 elements are inert gases, meaning they don't react with any other elements. Elements that occupy the same column on the periodic table (called a "group") have identical valence electron configurations and consequently behave in a similar fashion chemically.
#Periodic table full
As an example, elements in Group 8A (or VIIIA) all have a full set of eight electrons in the highest-energy orbital, according to chemist William Reusch, on his webpage at Michigan State University (opens in new tab). The columns, or groups, on the periodic table represent the atomic elements that have the same number of valence electrons, or those electrons in the outermost orbital shell.

(Atoms have protons and neutrons in their nucleus, and surrounding that, they have their electrons arranged in orbitals, where an atomic orbital is a math term that describes the location of an electron as well as its wave-like behavior.)įor instance, period 1 includes elements that have one atomic orbital where electrons spin period 2 has two atomic orbitals, period 3 has three and so on up to period 7. The horizontal rows on the periodic table are called periods, where each period number indicates the number of orbitals for the elements in that row, according to Los Alamos National Laboratory (opens in new tab).
